- The newly renamed PMOS condition affects 1 in 10 women of reproductive age, with far-reaching effects on metabolism, mental health, and hormonal balance.
- The redefinition of PCOS to PMOS validates years of dismissed symptoms, including insulin resistance, hirsutism, and chronic fatigue.
- Diagnostic delays averaging six years have long plagued women seeking care for PMOS, and a global medical consensus aims to address this issue.
- The shift from PCOS to PMOS reflects a scientific recalibration, emphasizing the broader metabolic and endocrine disruptions at play.
- The new name aims to dismantle the stigma surrounding women seeking care for PMOS, promoting a more accurate understanding of the condition.
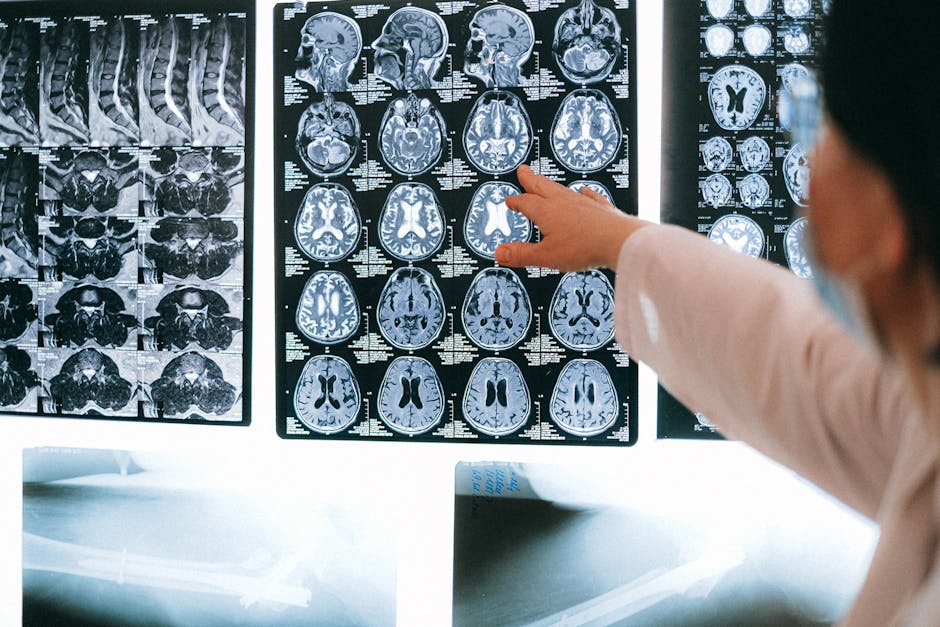
One in ten women of reproductive age lives with what was once simplistically labeled polycystic ovary syndrome (PCOS)—a condition long reduced to ovarian cysts and irregular periods, despite its far-reaching effects on metabolism, mental health, and hormonal balance. Now redefined as polyendocrine metabolic ovarian syndrome, or PMOS, this condition is finally being recognized not as a gynecological anomaly but as a complex, multisystem endocrine disorder. For millions, the change is more than semantic: it validates years of dismissed symptoms, from insulin resistance and hirsutism to chronic fatigue and depression. As one patient put it, “I still want to scream—because no one believed me for over a decade.” The global medical consensus, formalized in a 2026 joint statement by the World Health Organization and the Endocrine Society, aims to dismantle diagnostic delays that average six years and reduce the stigma that has long shadowed women seeking care.
A Name That Reflects Biological Reality
The shift from PCOS to PMOS is not merely a rebranding but a scientific recalibration. For decades, the term “polycystic ovaries” emphasized a single anatomical feature visible on ultrasound, leading both clinicians and patients to overlook the broader metabolic and endocrine disruptions at play. In reality, not all patients exhibit ovarian cysts, yet they suffer from the same insulin dysregulation, elevated androgens, and chronic inflammation. By embedding “polyendocrine” and “metabolic” in the new name, PMOS more accurately captures the condition’s systemic nature, aligning it with disorders like type 2 diabetes and metabolic syndrome. This reframing, experts say, will encourage earlier screening for cardiovascular risk, diabetes, and mental health support—interventions long under-prioritized in conventional PCOS management.
Voices from the Frontlines of Diagnosis
Patient advocacy played a crucial role in driving the name change. Across social media, support groups, and clinical testimony, women have shared stories of being labeled “lazy” for weight gain linked to insulin resistance, or “hoverreacting” when reporting mood swings and brain fog. Many reported being prescribed birth control pills as a catch-all solution, while underlying metabolic dysfunction went unaddressed. “I was told my acne and hair loss were just ‘hormonal’ and to ‘lose weight,’” said Maria Tran, a 34-year-old teacher from Melbourne. “It took five doctors and three miscarriages before someone tested my HbA1c and fasting insulin.” The new PMOS framework mandates a broader diagnostic panel, including glucose tolerance, lipid profiles, and thyroid function, ensuring a more holistic assessment from the outset.
Scientific Rationale Behind the Reclassification
Mounting research over the past two decades has demonstrated that PMOS is not confined to the ovaries but involves dysregulation across multiple endocrine glands, including the pancreas, adrenal glands, and hypothalamus. A landmark 2024 review in Nature Reviews Endocrinology concluded that insulin resistance is present in up to 70% of cases, regardless of body weight, and is a primary driver of hyperandrogenism. Furthermore, epigenetic and inflammatory markers suggest a chronic low-grade inflammatory state, linking PMOS to increased risks of non-alcoholic fatty liver disease, sleep apnea, and depression. The new diagnostic criteria, published by the International Consortium on PMOS, emphasize a phenotype-based approach over imaging alone, integrating clinical, biochemical, and metabolic markers to improve accuracy and reduce disparities in care, particularly among Black, South Asian, and Indigenous populations who are disproportionately affected.
Implications for Healthcare Systems and Patients
The reclassification of PCOS as PMOS has far-reaching implications for public health policy and clinical practice. Health systems will need to update diagnostic guidelines, train providers in metabolic screening, and expand access to multidisciplinary care teams including endocrinologists, dietitians, and mental health professionals. Insurance providers are being urged to cover comprehensive testing and long-term management, rather than episodic symptom control. For patients, the change offers the potential for earlier diagnosis, personalized treatment plans, and reduced psychological burden. However, challenges remain: global implementation will require significant investment, and there is a risk of confusion during the transition period, especially in regions with limited healthcare infrastructure.
Expert Perspectives
While the renaming has been widely praised, some clinicians urge caution. Dr. Lena Patel, an endocrinologist at Johns Hopkins, warns that “while PMOS better reflects the science, we must ensure the new label doesn’t further medicalize normal hormonal variation in women.” Others, like Dr. Kwame Osei of the African Endocrine Society, emphasize that “the real test is whether this change reaches women in rural clinics who have never even heard of PCOS, let alone PMOS.” Patient advocates, meanwhile, see empowerment in the shift: “This isn’t just a new name,” said advocacy leader Fatima Nkosi. “It’s recognition that our bodies were speaking all along—we just needed medicine to listen.”
Looking ahead, the success of the PMOS reclassification will depend on global coordination, patient education, and equitable access to care. Clinical trials are already underway to test targeted therapies for insulin sensitization and inflammation modulation. As the medical community adopts the new framework, the hope is that no woman will have to wait years for a diagnosis—or feel the need to scream just to be heard.
Source: The Guardian